論文リスト
- 学術論文:2024 | 2023 | 2022 | 2021 | 2020 | 2019 | 2018 | 2017 | 2016 | 2015 | 2014 | 2013 | 2012 | 2011 | 2010 | 2009 | 2008 | 2007 | 2006 | 2005 | 2004 | 2003 | 2002
- 総説・著書
学術論文 2009
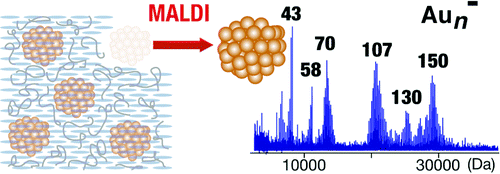
Magic Numbers of Gold Clusters Stabilized by PVP
Hironori Tsunoyama and Tatsuya Tsukuda*
J.
Am. Chem. Soc., 131, 18216-18217 (2009). ![]()
Size Determination of Gold Clusters by Polyacrylamide Gel Electrophoresis in a Large Cluster Region
Keisaku Kimura*, Nobuyuki Sugimoto, Seiichi Sato,
Hiroshi Yao, Yuichi Negishi, and Tatsuya Tsukuda
J.
Phys. Chem. C, 113, 14076-14082 (2009). ![]()
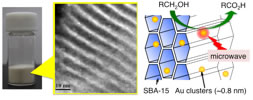
Preparation of ~1 nm Gold Clusters Confined within Mesoporous Silica and Microwave-Assisted Catalytic Application for Alcohol Oxidation
Yongmei Liu, Hironori Tsunoyama, Tomoki Akita, and
Tatsuya Tsukuda*
J.
Phys. Chem. C, 113, 13457-13461 (2009). ![]()
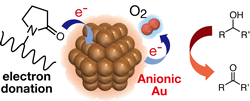
Effect of Electronic Structures of Au Clusters Stabilized by Poly(N-vinyl-2-pyrrolidone) on Aerobic Oxidation Catalysis
Hironori Tsunoyama, Nobuyuki Ichikuni, Hidehiro
Sakurai, Tatsuya Tsukuda*
J.
Am. Chem. Soc., 131, 7086-7093 (2009). ![]()
Aerobic Oxygenation of Benzylic Ketones Promoted by Gold Nanocluster Catalyst
Hidehiro Sakurai*, Ikuyo Kamiya, Hiroaki Kitahara,
Hironori Tsunoyama, Tatsuya Tsukuda
SYNLETT,
2, 0245-0248 (2009). ![]()
