論文リスト
- 学術論文:2024 | 2023 | 2022 | 2021 | 2020 | 2019 | 2018 | 2017 | 2016 | 2015 | 2014 | 2013 | 2012 | 2011 | 2010 | 2009 | 2008 | 2007 | 2006 | 2005 | 2004 | 2003 | 2002
- 総説・著書
学術論文 2003
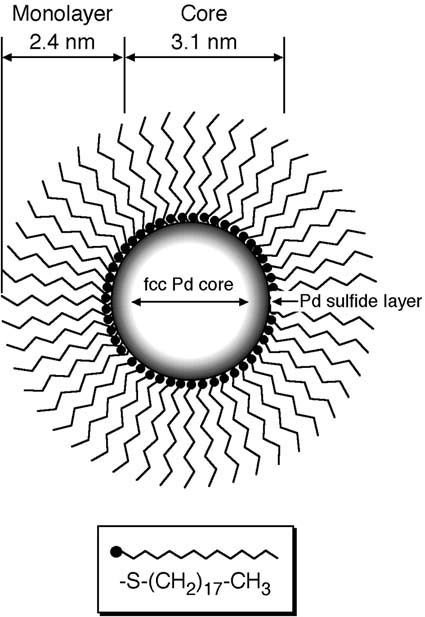
EXAFS Study on Interfacial Structure between Pd Cluster and n-Octadecanethiolate Monolayer: Formation of Mixed Pd-S Interlayer
H. Murayama, N. Ichikuni, Y. Negishi, T. Nagata,
and T. Tsukuda*
Chem. Phys. Lett., 376,
26-32 (2003). ![]()
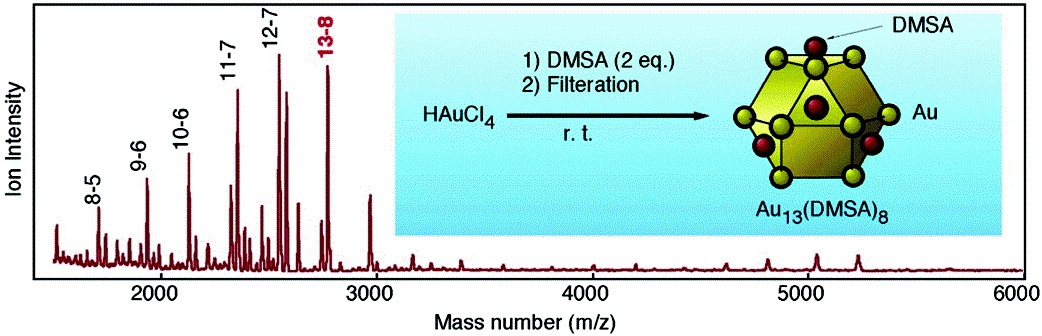
One-Pot Preparation of Subnanometer-Sized Gold Clusters via Reduction and Stabilization by meso-2,3-Dimercaptosuccinic Acid
Y. Negishi and T. Tsukuda*
J.
Am. Chem. Soc. (communications), 125, 4046-4047
(2003). ![]()
Self-Assembly of Si Clusters into Single Crystal Arrangements: Formation of Si10 Cluster Crystals
S. Sato, N. Yamamoto, K. Nakanishi, H. Yao, K.
Kimura, Y. Negishi, and T. Tsukuda
Jpn.
J. Appl. Phys., 42, L616-L618 (2003). ![]()
