論文リスト
- 学術論文:2024 | 2023 | 2022 | 2021 | 2020 | 2019 | 2018 | 2017 | 2016 | 2015 | 2014 | 2013 | 2012 | 2011 | 2010 | 2009 | 2008 | 2007 | 2006 | 2005 | 2004 | 2003 | 2002
- 総説・著書
学術論文 2007
Thermal and photochemical reactivity of oxygen atoms on gold nanocluster surfaces
T. Matsumoto, P. Nickut, H. Tsunoyama, K.
Watanabe, T. Tsukuda, K. Al-Shamery, Y. Matsumoto*
Surf.
Sci., 601, 5226-5231 (2007). ![]()
Deposition and Fabrication of Alkanethiolate Gold Nanocluster Films on TiO2(110) and the Effects of Plasma Etching
T. Matsumoto, P. Nickut, T. Sawada, H.
Tsunoyama, K. Watanabe, T. Tsukuda, K. Al-Shamery, Y. Matsumoto*
Surf.
Sci., 601, 5121-5126 (2007). ![]()
Thermosensitive Gold Nanoclusters Stabilized by Well-Defined Vinyl Ether Star Polymers: Reusable and Durable Catalysts for Aerobic Alcohol Oxidation
S. Kanaoka, N. Yagi, Y. Fukuyama, S. Aoshima,* H.
Tsunoyama, T. Tsukuda, and H. Sakurai
J.
Am. Chem. Soc. (communications), 129,
12060-12061 (2007). ![]()
Origin of Magic Stability of Thiolated Gold Clusters: A Case Study on Au25(SC6H13)18
Y. Negishi, N. K. Chaki, Y. Shichibu, R. L.
Whetten, and T. Tsukuda*
J.
Am. Chem. Soc. (communications), 129,
11322-11323 (2007). ![]()
Synthesis of Normal and Inverted Gold-Silver Core-Shell Architectures in β-Cyclodextrin and Their Applications in SERS
S. Pande, S. K. Ghosh, S. Praharaj, S. Panigrahi,
S. Basu, S. Jana, A. Pal, T. Tsukuda, and T. Pal*
J.
Phys. Chem. C, 111, 10806-10813 (2007). ![]()
Electronic Structure of Dendrimer-encapsulated Au Nanocluster
M. Imamura,* T. Miyashita, A. Tanaka, H. Yasuda, Y. Negishi, T. Tsukuda
Eur.
Phys. J. D, 43, 233-236 (2007). ![]()
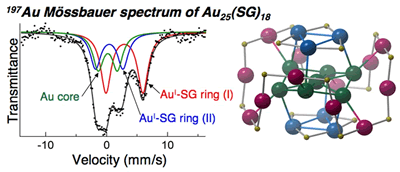
Thiolate-Induced Structural Reconstruction of Gold Clusters Probed by 197Au Mössbauer Spectroscopy
K. Ikeda, Y. Kobayashi, Y. Negishi, M. Seto, T.
Iwasa, K. Nobusada, T. Tsukuda,* and N. Kojima*
J.
Am. Chem. Soc. (communications), 129, 7230-7231
(2007). ![]()
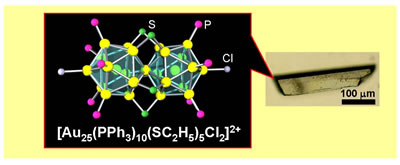
Biicosahedral Gold Clusters [Au25(PPh3)10(SCnH2n+1)5Cl2]2+ (n=2-18): a Stepping Stone to Cluster-Assembled Materials
Y. Shichibu, Y. Negishi, T. Watanabe, N. K. Chaki,
H. Kawaguchi, and T. Tsukuda*
J.
Phys. Chem. C (Letters), 111, 7845-7847 (2007).
![]()
Lewis Acid Character of Zero-valent Gold Nanoclusters under Aerobic Conditions: Intramolecular Hydroalkoxylation of Alkenes
I. Kamiya, H. Tsunoyama, T. Tsukuda, and H. Sakurai*
Chem.
Lett., 36, 646-647 (2007). ![]()
Effect of Ag-Doping on the Catalytic Activity of Polymer-Stabilized Au Clusters in Aerobic Oxidation of Alcohol
N. K. Chaki, H. Tsunoyama, Y. Negishi, H. Sakurai,
T. Tsukuda*
J.
Phys. Chem. C (Letters), 111, 4885-4888 (2007).
![]()
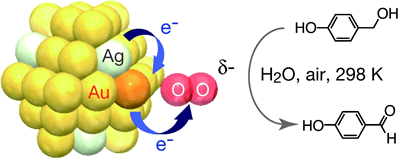
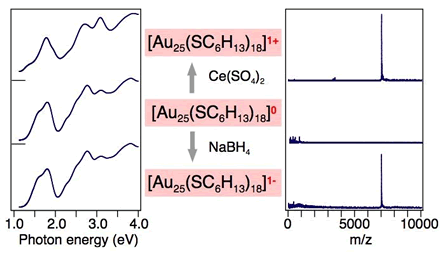
Extremely High Stability of Glutathionate-Protected Au25 Clusters Against Core Etching
Y. Shichibu, Y. Negishi, H. Tsunoyama, M. Kanehara,
T. Teranishi, and T. Tsukuda*
Small(communications),
3, 835-839 (2007). ![]()
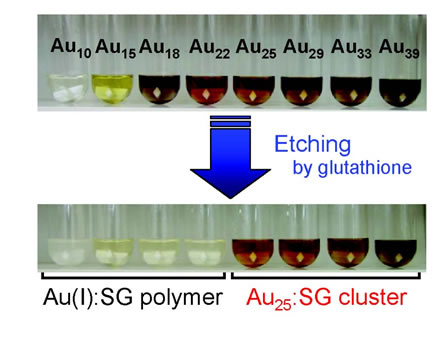
Formation of Alkanethiolate-Protected Gold Clusters with Unprecedented Core Sizes in the Thiolation of Polymer-Stabilized Gold Clusters
H. Tsunoyama, P. Nickut, Y. Negishi, K. Al-Shamery,
Y. Matsumoto, T. Tsukuda*
J.
Phys. Chem. C, 111, 4153-4158 (2007). ![]()
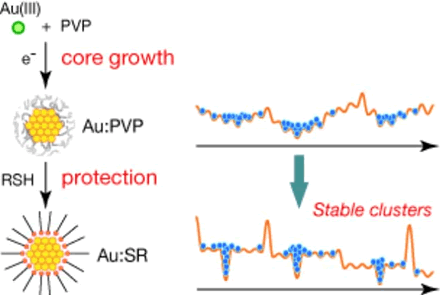
Synthetic Application of PVP-stabilized Au Nanocluster Catalyst to Aerobic Oxidation of Alcohols in Aqueous Solution under Ambient Conditions
H. Tsunoyama, T. Tsukuda, H. Sakurai*
Chem.
Lett., 36, 212-213 (2007). ![]()
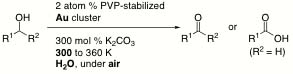
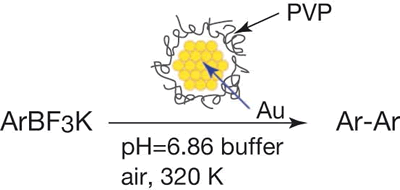
Oxidative Homo-coupling of Potassium Aryltrifluoroborates Catalyzed by Gold Nanocluster under Aerobic Conditions
H. Sakurai*, H. Tsunoyama, and T. Tsukuda
J. Organomet. Chem., 692,
368-374 (2007). ![]()