New Reactions Utilizing Carbon Dioxide
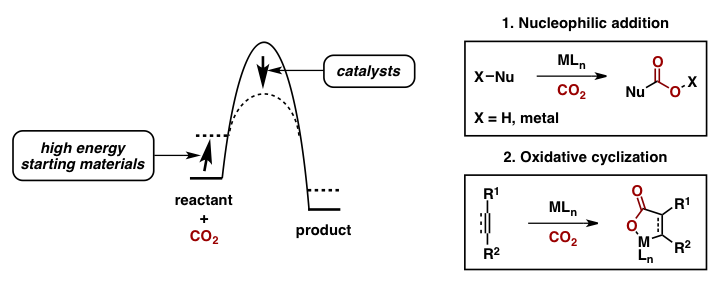
The chemical utilization of carbon dioxide (CO2) as a C1 feedstock to access value added materials is attractive since it is a safe, inexpensive, and readily available gas. However, due to its intrinsic kinetic and thermodynamic stability, the use of carbon dioxide is limited. In order to overcome these barriers, the strategic use of highly energetic starting materials and/or intermediates, in conjugation with a catalyst, allows for the development of useful synthetic methodologies that can incorporate CO2 into complex organic molecules.
Topics
- Multicomponent Coupling Reactions with CO2
- Base-Mediated Carboxylation of Indole and Pyrrole Derivatives
Multicomponent Coupling Reactions with CO2
Comment
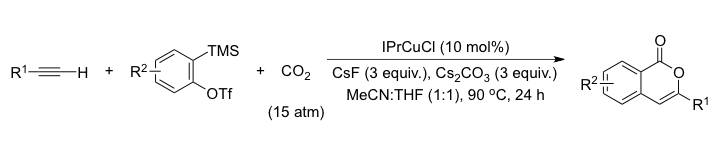
A multicomponent coupling reaction between in situ generated benzynes, terminal alkynes, and CO2 to prepare isocoumarins was developed. The key to this CO2-incorporating reaction was the use of a versatile N-heterocyclic carbene-copper complex that enabled the various organic transformations within the three-component coupling reaction.
Access to paper
- Synthesis of Isocoumarins Through Three-Component Couplings of Arynes, Terminal Alkynes, and Carbon Dioxide Catalyzed by an NHC-copper complex,
- W.-J. Yoo, T. V. Q. Nguyen, S. Kobayashi,
- Angew. Chem. Int. Ed., 53, 10213-10217 (2014). DOI: 10.1002/anie.201404682
Base-Mediated Carboxylation of Indole and Pyrrole Derivatives
Comment
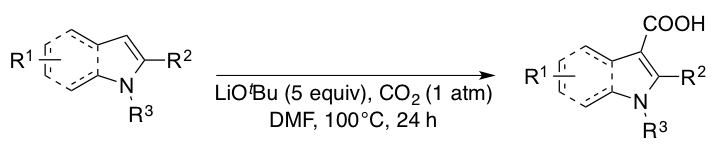
Unprotected indoles and pyrroles were found to undergo base-mediated carboxylation reactions under ambient pressure of carbon dioxide. The key for the reaction was found to be the use of a large excess of LiOtBu as a base to suppress the undesired decarboxylation side reaction.
Access to papers
- Base-Mediated Carboxylation of Unprotected Indole Derivatives with Carbon Dioxide,
- W.-J. Yoo, M. Guiteras Capdevila, X. Du, S. Kobayashi,
- Org. Lett., 14, 5326-5329 (2012). DOI: 10.1021/ol3025082
- Lithium tert-Butoxide-Mediated Carboxylation Reactions of Unprotected Indoles and Pyrroles with Carbon Dioxide,
- W.-J. Yoo, T. V. Q. Nguyen, M. Guiteras Capdevila, S. Kobayashi,
- Heterocycles, 90, 1196-1204 (2015). DOI: 10.3987/COM-14-S(K)94