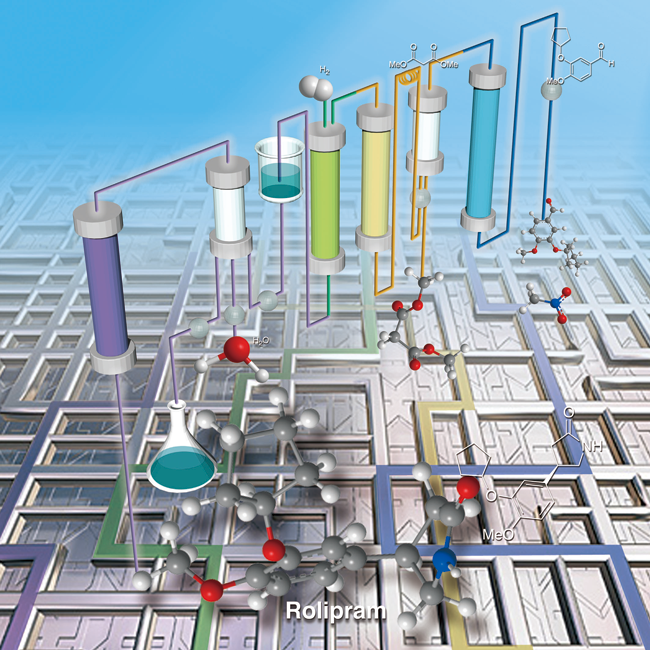
Flow "Fine" Synthesis
In the area of fine chemical production, chemical products such as medicines, agrichemicals and functional materials are often synthesized in batch systems. However, those methods are not suitable for view point of GSC (Green Sustainable Chemistry) due to the production of much amount of chemical wastes.
On the other hand, synthesizes through flow systems are usually preferred in bulk chemical production and petrochemistry i.e. Haber-Bosch ammonia production. We are considering that these flow systems should be applied for fine chemical production, especially drugs.
There are many advantages in flow systems compering to batch systems. Firstly, this system provides high economic efficiency and easy production control. Also it can save space, time and energy. Secondly, it is safe. Finally, we could prepare the multi-step reaction systems just connecting different flows, this causes avoidance of isolation and purification of intermediates. These advantages fully meet the criteria of GMP (Good Manufacturing Practice) policy. In recent, US FDA commented“right now, manufacturing experts from the 1950s would easily recognize the pharmaceutical manufacturing processes of today. It is predicted that manufacturing will change in the next 25 years as current manufacturing practices are abandoned in favor of cleaner, flexible, more efficient continuous manufacturing.”
We are considering that the above flow chemistry needs strategically new idea of protocols to achieve high atom efficiency, and we are now working on the development of new chemical reactions and effective heterogeneous/solid catalysts suitable for the continuous flow syntheses.
Reviews
- Flow “Fine” Synthesis: High Yielding and Selective Organic Synthesis by Flow Methods
- S. Kobayashi,
- Chem. Asian J., Early View , (2015). DOI: 10.1002/asia.201500916
- Asymmetric Carbon-Carbon Bond Formation under Continuous Flow Conditions with Chiral Heterogeneous Catalysts,
- T. Tsubogo, T. Ishiwata, S. Kobayashi,
- Angew. Chem. Int. Ed., 52, 6590-6604 (2013). DOI: 10.1002/anie.201210066 more details