Platelets are an important type of anucleate cell found in blood, which are derived from megakaryocytes in the bone marrow. Their main role is to prevent bleeding by initiating hemostatic reactions that result in clot formation. The activity of platelets is regulated by various stimuli, which interact with different receptors on their surface to trigger a series of downstream events that induce their structural and functional changes, leading to hemostasis. This complex process involves a vast number of surface receptors and agonists, and occurs simultaneously with coagulation cascades involving blood cells and endothelial cells that are triggered by tissue factor, leading to fibrin formation via thrombin.
In addition to their role in hemostasis, platelets are also involved in various pathological processes such as thrombosis, atherosclerosis, inflammation, coronary artery disease, diabetes mellitus, tumor growth, cancer metastasis, and COVID-19. Platelets interact directly or indirectly with other types of cells such as leukocytes, vascular endothelial cells, and tumor cells, using diverse mechanisms. For example, platelets protect circulating tumor cells (CTCs) from shear forces and cytolysis by natural killer cells by encasing CTCs in thrombi.
The study of platelets and their complex physiological and pathological functions requires advanced image-based analysis techniques, such as platelet morphometry. Such techniques also aid in the development of more effective clinical strategies. To this end, we have collaborated with clinicians from the University of Tokyo Hospital to develop an innovative tool that utilizes high-throughput optical imaging and deep learning to advance platelet morphometry beyond traditional methods. This new approach, which we call "intelligent platelet morphometry," provides a new set of capabilities and sheds light on previously unexplored areas of platelet analysis. This promising field has the potential to revolutionize diagnostics, pharmacometrics, and therapeutics.
References
- Y. Zhou, A. Isozaki, A. Yasumoto, T. Xiao, Y. Yatomi, C. Lei, and K. Goda, "Intelligent platelet morphometry", Trends in Biotechnology 39, 978 (2021)
- Y. Deng, J. A. Duque, C. Su, Y. Zhou, M. Nishikawa, T. Xiao, Y. Yatomi, H. W. Hou, and K. Goda, "Understanding stenosis-induced platelet aggregation on a chip by high-speed optical imaging", Sensors and Actuators B: Chemical 356, 131318 (2022)
- M. Nishikawa, H. Kanno, Y. Zhou, T. Xiao, T. Suzuki, Y. Ibayashi, J. Harmon, S. Takizawa, K. Hiramatsu, N. Nitta, R. Kameyama, W. Peterson, J. Takiguchi, M. Shifat-E-Rabbi, Y. Zhuang, X. Yin, A. H. M. Rubaiyat, Y. Deng, H. Zhang, S. Miyata, G. K. Rohde, W. Iwasaki, Y. Yatomi, and K. Goda, "Massive image-based single-cell profiling reveals high levels of circulating platelet aggregates in patients with COVID-19", Nature Communications 12, 7135 (2021)
- K. C. M. Lee, J. Guck, K. Goda, and K. K. Tsia, "Deep biophysical cytometry: prospects and challenges", Trends in Biotechnology 39, 1249 (2021)
- Y. Zhou, A. Yasumoto, C. Lei, C.-J. Huang, H. Kobayashi, Y. Wu, S. Yan, C.-W. Sun, Y. Yatomi, and K. Goda, "Intelligent classification of platelet aggregates by agonist type", eLife 9, e52938 (2020)
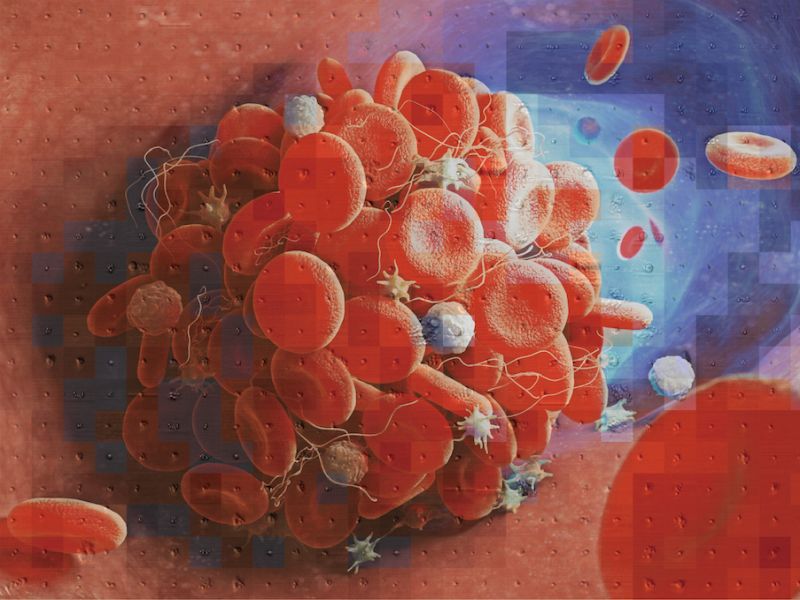
Intelligent platelet morphometry
- Field leader: Yuqi Zhou
- Funding: JSPS Core-to-Core Program, AMED Interdisciplinary Cutting-Edge Research, KAKETSUKEN
- Collaboration: Clinical Laboratory at the University of Tokyo Hospital